- Thermophysics
- /
- Heat
- /
- Phase transitions
"Solid, liquid or gas? Substance, temperature and pressure?"
Phase Transitions
The substances can be in three physical states: solid, liquid or gaseous, see figure.

For a phase change to occur, it is necessary to provide (or remove) an amount of heat \(Q\) of the substance. During the phase change, the energy received (or given) is used to change the organization of the molecules, so that, at this time, there is no variation in temperature.
The most important definitions are:- Pure Substance
- It is any matter whose melting and boiling occurs at constant temperature. It is also referred simply as substance.
- Latent Heat of Phase Change \((L)\)
- It is the amount of heat \(L\) required to change the phase of a unit mass of the substance of interest. That is, for a mass system \(m\) that is already in the phase change temperature and a constant pressure, the energy required for the system to change of phase is: $$Q = L \times m.$$
- Internal Energy
- When a substance changes from solid to liquid, liquid to gas or solid to gas, the internal energy of the body increases even in the case where the temperature does not change. During the inverse transformation, the body's internal energy decreases.
Phases Diagram
The different stages and their coexistences can be best viewed in a diagram \(P \times T\) , see figure below.
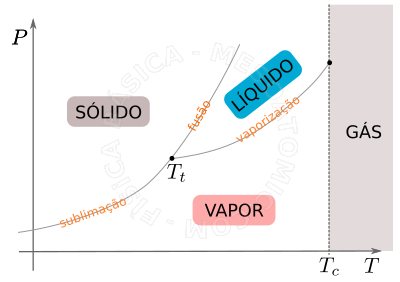
- Triple Point
- It is the only point where all states can coexist in equilibrium stages for a given temperature value (\(T_t\)) and specific pressure.
- The Critical Temperature \((T_c)\)
- Above the critical temperature it is not possible to liquefy the substance through a simple isothermal compression, independent of how greater the pressure may be, i.e., above the critical temperature the substance is called gas.
- Evaporation
- It is the phenomenon of transition from liquid to gas. Molecules that are in the liquid-gas interface start belong to the gas. It is important to note that:
- Evaporation is a phenomenon that occurs at any temperature.
- The higher the temperature the greater will be the net rate of evaporation.
- When there is air flow (wind) on the surface of a liquid, the evaporation rate increases.
- The evaporation rate of a liquid depends on the area of the liquid surface.
- A steam in equilibrium with its liquid at a given temperature exerts maximum vapor pressure.
LATENT HEAT OF TRANSFORMATION FUSION BOILING Substance Score Heat Score Heat \(IS\) K KJ / kg K KJ / kg Hydrogen 14.0 58.0 20.3 455 Mercury 234 11.4 630 296 Water 273 333 373 2256 Lead 601 23.2 2017 858 Silver 1235 105 2323 2336 Copper 1356 207 2868 4730
Laws of Melting and Solidification
The melting and solidification of a pure substance depend only on the pressure and nature of the substance. That is, if the substance is pure and the pressure on it is constant, then \(T_{Melt} = a\) and \(T_{Solid} = b,\) where \(a\) and \(b\) are constant.
During melting, or solidification, the pure substance has some volume change.
Water
Water is one of the fundamental elements for life as we know it, and understanding their properties is crucial.-
Latent Heat Value (cal / g) Fusion ( \(L_F\) ) 80 Solidification ( \(L_S\) ) -80 Vaporization ( \(L_V\) ) 540 Condensation ( \(L_C\) ) -540 -
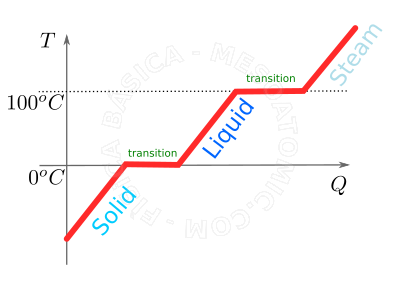
Curve of heating water under 1 atmosphere of pressure. This outlines the different phases the water undergoes when heated, i.e., as it receives heat. Initially, at a sufficiently low temperature, the water is in solid form, i.e., in the form of ice. As it receives heat, its temperature rises until it reaches \(0^oC\). When ice reaches \(0^oC\), if it continues to receive heat, its temperature still does not increase, what happens is a phase transition. At this temperature, the heat received will serve to transform the ice in liquid and not to increase the temperature (this is illustrated by the first plateau in the figure). Once all the ice turned into liquid at \(0^oC\), if it get more heat, again the system temperature will grow until reaching \(100^oC\), where again a phase transition occurs. -
The Kelvin Scale Reference
The temperature of the triple point is an ideal reference for temperature ranges, requiring very specific conditions of pressure and temperature to get to this point. This provides a benchmark which results in a more accurate scale. Therefore, the Kelvin scale is defined as a linear temperature scale which begins at \(0K\), the absolute zero, and passes through \(273.16K\) at the triple point of water.
