- Electromagnetism
- /
- Electrostatic
- /
- Electric charges
The electric charge is a physical quantity that determines the intensity of electrostatic interactions.
Electrical charge
- Types of electric charges
- There are only two types, positive and negative.
- Du Fay's Principle
- Two bodies electrified with different charges (one positive and one negative) attract each other. Bodies with the same type of charges (both positive or both negative) repel each other.
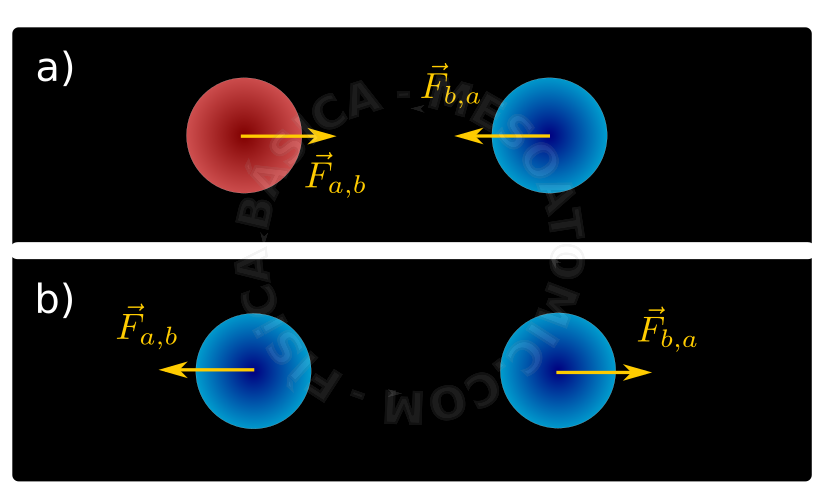
Illustration of forces between electrified bodies. a) oppositely charged bodies attract each other and b) bodies with the same charges repel each other. - Conservation of Electric Charges
- The total amount of electrical charges does not change in any process performed within an isolated system.
- Quantization of Electric Charge
- All charges that are observed in nature are equal or multiple of the elementary charge \(e\).
- Elementary Electric Charge
- Electron: \(-e\) (negative elementary charge)
Proton: \(+e\) (positive elementary charge)
where \(e = 1,6 \times 10^{-19} C\) .
The electric charge unit in \(IS\) is the Coulomb (\(C\)).
The net amount of charge\((Q)\)
The net charge a body possesses is equal to the number of excess of elementary charges (positive or negative) times the elementary charge, i.e., \begin{equation} Q = ne \end{equation} where \(n\) is the number of electrons transferred or received, and \(e\) is the value of the elementary charge.
Every atom is electrically neutral if its number of protons is equal to the number of electrons.
